Getting My Medicinal Cannabis Agency – Cannabidiol (Cbd) Products To Work
If an item is meant to influence the structure or function of the body, or to detect, heal, alleviate, treat or stop disease, it is a medication, or perhaps both a cosmetic as well as a medicine, even if it influences the look. (See Inquiry # 3 for even more information concerning drugs.) FDA can act if it knows that an ingredient or aesthetic product is risky to customers.
Those factors consist of, amongst various other points, company sources as well as the hazard to the public wellness. FDA also might seek advice from with its government and also state companions in making choices concerning whether to initiate a government enforcement action.
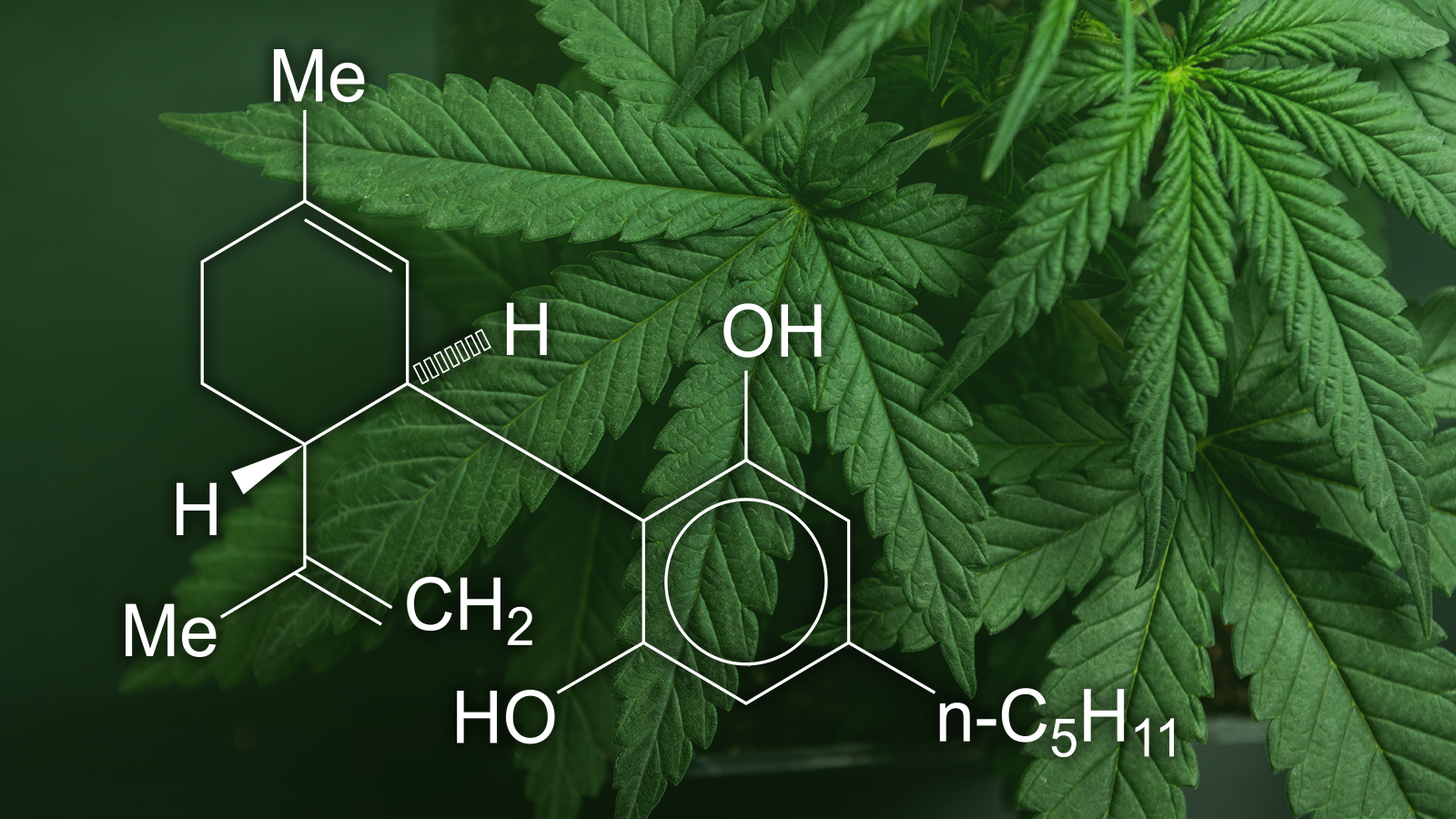
21 U.S.C.
A. To conduct clinical perform that can lead to an approved new accepted, including research consisting of research study making use of products such as cannabis, researchers need to work with the FDA and submit and also IND application to the Center for Drug Evaluation medicine Assessment As well asCDER). The IND application procedure offers scientists a path to follow that includes routine communications with the FDA to sustain reliable medicine advancement while shielding the people that are registered in the trials.
This includes: a registration provided by the DEA; obtaining the marijuana for research study from NIDA, within the National Institutes of Health And Wellness, or an additional DEA-registered resource; and testimonial by the FDA of the IND or INAD application and research procedure. Additionally: For a Schedule I controlled compound under the CSA, DEA offers scientists with detective as well as method enrollments and also has Arrange I-level protection demands at the site marijuana will be studied.
All about Research Shows That, Genetically, High-cbd “Hemp” Plants Are …
Based on the results acquired in researches conducted at the IND phase, enrollers might send a marketing application for formal approval of the medication. A. No. The FDA thinks that scientifically valid study performed under an IND application is the most effective means to establish what individuals could benefit from using drugs derived from marijuana.
Companies who develop drugs and biologics, additionally called enrollers, can offer details regarding whether their drug/biologic is thought about an eligible investigational medication under RTT and if they have the ability to https://wayofleaf.com/accessories/bongs offer the drug/biologic under the RTT Act. A. We recognize that moms and dads are looking for therapies for their youngsters’s clinical conditions.
Caregivers and people can be certain that FDA-approved drugs have been very carefully assessed for safety, efficiency, and also top quality, as well as are monitored by the FDA once they get on the market. The FDA proceeds to sustain noise, scientifically-based research study into the medical uses of drug items containing cannabis or cannabis-derived compounds, and also will remain to collaborate with business fascinated in bringing safe, efficient, as well as high quality products to market.
About Cbd & Diabetes – Ada
The American College of Obstetricians as well as Gynecologists (ACOG) recommends that women that are expectant or considering pregnancy should be urged to cease marijuana usage. In addition, ACOG notes that there are not enough data to evaluate the effects of marijuana usage on nursed babies; as a result, marijuana use is discouraged when nursing.
A. The FDA has actually accepted Epidiolex, which consists of a cleansed type of the medication compound CBD, for the treatment of seizures connected with Lennox-Gastaut syndrome or Dravet disorder in people 1 years of age and also older. It has actually likewise authorized Epidiolex for the therapy of seizures connected with tuberous sclerosis complicated in clients 1 year old or older.
Controlled professional trials checking the safety and security and also effectiveness of a medication, together with mindful evaluation through the FDA’s medication authorization process, is the most ideal method to bring cannabis-derived treatments to individuals. Due to the sufficient and well-controlled scientific researches that sustained this authorization, as well as the guarantee of making quality requirements, prescribers can have self-confidence in the drug’s uniform toughness as well as consistent delivery that support appropriate dosing needed for treating clients with these facility and significant epilepsy disorders.
Some Ideas on Medicinal Cannabis Agency – Cannabidiol (Cbd) Products You Should Know
With the exemption of products such as the hemp seed active ingredients gone over in Question # 12, which have been examined for safety, it is necessary to protect children from accidental intake of cannabis as well as cannabis-containing products. FDA advises that these items are maintained out of reach of children to reduce the threat of unintentional consumption.
